
Today’s pharmaceutical companies face economic and regulatory pressures, accelerating costs, technological and scientific changes, patent expirations and the need to constantly adapt their business models, particularly to comply with healthcare reform.
To meet these challenges, pharmaceutical companies must reduce costs, become more agile and improve speed to market while ensuring compliance with complex regulations. Doing so while positioning your company to scale and maximize financial performance is no small feat. It takes an unrelenting commitment to continuous optimization of core business processes, communication and collaboration across your organization.
But that’s not all. You also need to have the right enterprise resource management (ERP) and customer relationship management (CRM) systems in place. Increasingly, this means deploying a unified ERP/CRM solution, a solution with tight integration for end-to-end operational control that is specifically designed for the pharmaceutical industry. A unified, industry-specific solution gives you the level of visibility, control, and compliance you need to scale and respond to market and regulatory requirements, while controlling costs and improving your margins.
This white paper discusses the unique challenges of the pharmaceutical industry and the impact of meeting those challenges on core back- and front-office business processes. We’ll take a close look at today’s software requirements for pharmaceutical companies and the benefits of deploying a unified, industry-specific ERP/CRM solution.
The global pharmaceuticals market is worth $300 billion a year, a figure expected to rise to $400 billion within three years.
World Health Organization
Healthcare reform isn’t the only driver of far-reaching change in the U.S. life sciences industry. There are numerous trends and issues that pharmaceutical companies face in today’s marketplace. These challenges play out in crucial ways within your business processes:
accelerating the development of innovative pharmaceuticals Pharmaceutical companies are under tremendous pressure to create new therapies that are clinically and economically better than the existing alternatives, and to provide real-world data to support any claims of superiority. To supplement in-house R&D, many pharmaceutical companies are outsourcing to clinical research organizations. This means the business processes around R&D must be integrated and provide visibility into innovation process.
driving growth and expansion The global pharmaceutical market is expanding, with increasing demand stemming from global population growth, increased age and people living more sedentary lives. To take advantage of the growth potential, pharmaceutical companies must be prepared for increasing product volume, market scope and suppliers, all of which will require optimizing key production, planning and operational business processes.
meeting regulatory requirements As one of the most highly regulated industries, life sciences companies are intimately familiar with regulations from the U.S. and foreign markets. The complex, shifting regulatory environment means that your business processes must be agile and adaptable to many ongoing changes required by these standards
managing complex supply chains and sales channels The pharmaceutical value chain is more complex than ever, including a mix of hospital, provider and physician networks together with outsourced or contract manufacturing, R&D and diverse suppliers. Alignment of supply and demand is a core benefit of an integrated CRM and ERP solution. It is critical to have the right tools and processes in place to aggregate sales forecasts and accurately track the pipeline across diverse sales channels globally. This capability provides the high-quality, demand-planning information required from the front office to improve planning and decision making and optimize inventory levels across the supply chain.
managing product quality Supply chain complexity is also a major challenge in managing product quality. It’s imperative for pharmaceutical companies to conquer the complexity of quality controls, corrective and preventive actions (CAPAs) and root cause analyses. That’s another reason why pharmaceutical companies require greater visibility into the supply chain – to ensure both quality control and compliance.
To survive and thrive, pharmaceutical companies must seek out a solution that unifies processes across their back and front offices for end-toend efficiency, visibility and performance. Unified processes to address growth, innovation, compliance and increased supply chain complexity require a specialized, unified ERP/CRM solution designed for life sciences.
... on the manufacturing side [of the healthcare supply chain], inventory visibility is key to many of the ongoing working capital initiatives and agility strategies. Distributors … see the importance of visibility in improving supply shortages faced throughout the year, and improving recall processes.
Gartner 2012
Unlike stand-alone ERP and CRM systems, a unified front- and back-office solution designed for pharmaceutical companies can better support end-to-end visibility and control of life sciences-specific business processes. This enables you to align the supply and demand chains, maximize financial performance, and comply with relevant regulations. Equally important is the ability to efficiently validate your solution per FDA regulations.
Unify Your Business Processes
Selecting an ERP/CRM solution designed specifically for pharmaceutical companies is critical to unifying your business processes across departments, locations and systems. Why? You need to enable data to flow automatically to and from front-and back-office operations to deliver end-to-end visibility and control. There are many cross-system business processes that your ERP/CRM solution should support, including:
While many solutions offer some degree of integration to support these processes, there are also special requirements for pharmaceutical companies that make the need for a life sciences-specific, unified ERP/CRM solution imperative, including:
pricing and promotions Information about volume purchase agreements, customer-specific pricing, and promotions needs to flow from your CRM to your ERP so that you can reconcile commission payments, costs and negotiated prices. Many companies manually input information from CRM into the ERP system, reducing productivity and hindering real-time change. Your ERP/CRM solution should enable a unified process, allowing automatic data transfer between the systems.
q-to-cash Support for an end-to-end, q-to-cash business process can help significantly improve your time to revenue. A unified process across your ERP/CRM solution automatically synchronizes customer, product, pricing, qs, orders, invoices, revenue recognition and collections information.
customer service Whether you’re engaging directly with patients or supporting doctors and hospitals, a direct view from the CRM to the ERP can improve customer service with more accurate and synchronized information about customers and sales. Giving your customer service representatives real-time access to this data lets them resolve questions faster and improve customer satisfaction.
quality management All reported, quality-related product complaints and non-conforming material reports (NCMRs) from production quality control tests must be analyzed by the quality assurance organization to assess whether further corrective action and preventative action (CAPA) is warranted. Key ERP/CRM capabilities that support this process include:
demand forecasting Seek out a solution that can provide detailed, global information on orders (planned versus actual) over time against trade agreement commitments to improve coordination among your sales, financial and manufacturing groups, as well as partners. Information captured in the CRM flows through to the ERP for production planning, while actual billings from ERP flow back to CRM for dashboard reporting.
automated internal controls Your unified ERP/CRM system should enable you to increase visibility to risks, improve related financial cycle and general IT internal controls, and reduce recurring costs for external auditor controls testing via automation. Key capabilities include:
Sunshine Act spend tracking For compliance with the Physician Payment Sunshine Act (Section 6002 of the Patient Protection and Affordable Care Act), your unified ERP/CRM solution should track payments and other transfers of value made from your company to other healthcare providers (HCPs). Reporting information can be shared with a centralized aggregate-spend solution to facilitate physician/vendor review, potential dispute resolution and formal reporting to the Centers for Medicare and Medicaid Services.
global compliance management Your unified ERP/CRM system should facilitate compliance with:
marketing return on investment Your pharmaceutical company can tie campaigns in the CRM to closed business in your ERP to track marketing return on investment, gain insight into customer behavior to optimize your campaigns and grow lifetime customer value.
channel sales workflow A unified ERP/CRM system allows the CRM to streamline the quoting and ordering process for your partners, while forecasting information for partner-driven deals is automatically transferred to the ERP system, creating more accurate production planning.
Critical ERP Capabilities for Pharmaceutical Companies
Look for the following back-office capabilities when choosing a new, industryspecific ERP system:
Must-Have CRM Capabilities for Pharmaceutical Companies
Pharmaceutical companies have unique marketing and compliance requirements that go beyond what a standard CRM system is built to handle.
Specifically, your CRM system will need the following capabilities:
To achieve your goals and compete with the rest of the pharmaceutical industry, you need a software/solution provider with documented success of helping its customers validate their systems. Your solution partner should have a proven, integrated implementation and validation methodology, automated testing tools, and other methods and services to ease the burden of validation and speed implementation.
Armanino brings together industry-leading ERP and CRM capabilities in one unified solution specifically designed for life sciences companies. With more than 20 years of experience developing solutions for the pharmaceutical industry, Armanino can provide full-service implementation support for your end-to-end business processes. We bring deep industry best-practice expertise to accelerate the deployment of a unified solution across your enterprise.
Tailored, Industry-Leading Technologies
As one of the largest ERP and CRM Gold Certified Microsoft Dynamics Partners in the country, Armanino is the only Microsoft VAR with comprehensive industry-specific application functionality designed for the life sciences industry. With a Microsoft Dynamics solution from Armanino, you gain the visibility you need for end-to-end process management of your enterprise, including customer-facing processes, management of the supply chain, production, HR, and consolidated financials. You can balance demand with production efforts, maintain optimum inventory levels, and meet customer expectations profitably. Figure 1 shows how the Microsoft Dynamics solution supports end-to-end, unified processes across the business.
Microsoft Dynamics brings together the broad range of Microsoft technology, making it easier for businesses to collaborate, connect, and deliver results. Microsoft Lync, SharePoint, and SQL Server BI tools provide embedded communications, collaboration, and analytics capabilities within a comprehensive ERP/CRM business software suite. Microsoft Dynamics CRM runs in Outlook, so users can move between tasks with greater ease and without disruption.
This pervasive interoperability drives productivity, creates a better user experience, reduces IT costs and complexity, and puts new Microsoft products into customers’ hands without headache. For example, it is easy for your IT department to support the Dynamics suite of solutions on the familiar Windows platform and SQL infrastructure they work with daily. Users can seamlessly move data between Microsoft Dynamics business applications and Microsoft Excel to empower ad-hoc analysis.
Ready-to-Use Templates
Armanino provides award-winning sales, implementation and support services for Microsoft Dynamics, including system implementation, integration, and customization, as well as technical support and IT outsourcing. Another unique aspect of an Armanino solution is our set of preconfigured templates for the life sciences industry. These templates reflect operations, accounting, and software configuration best practices to improve quality and reliability, lower costs, and accelerate implementation. Templates include quality management, revenue recognition and workflow.
Regulation and Compliance Solutions
The Armanino solution supports the relevant regulatory requirements with which pharmaceutical companies must comply. The unified Armanino ERP/CRM solution has built-in capabilities, such as quality control monitoring and overall quality management processes, sales restrictions for unqualified customers, and product and supply-chain transaction tracing.
Armanino also helps lessen the burden of FDA system validation for your solution. Based on Good Automated Manufacturing Practice (GAMP) best practices, our Accelerator for ERP software validation helps you create the objective evidence you need to show that your system functions per its intended use. The Accelerator includes validation templates, methodology, and documentation combined with comprehensive professional services to streamline and simplify the validation process for your implementation.
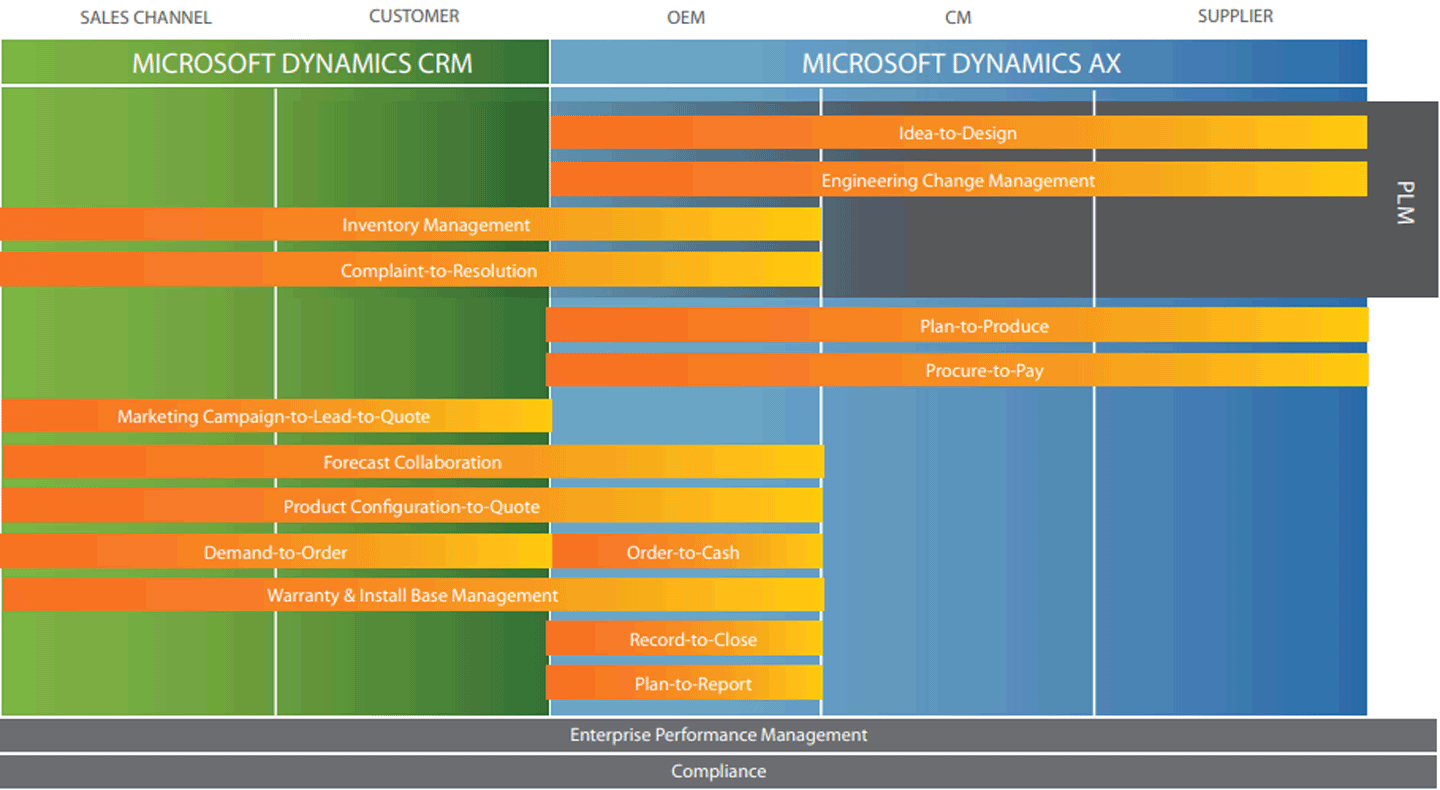
Microsoft Dynamics AX and Microsoft Dynamics CRM Process Support
Managing product quality despite increasing supply chain complexity, navigating the effects of patent cliffs and R&D pressures, complying with industry regulations – the list of challenges facing pharmaceutical companies continues to grow. Overcoming these issues is paramount for your business to survive and thrive. Therefore, you need to have the right infrastructure in place to support and manage complex, end-to-end business processes that span multiple departments, divisions and third parties. That infrastructure is a unified, industry-specific ERP/CRM solution.
Armanino’s Microsoft Dynamics ERP/CRM for Life Sciences solution gives you the level of visibility, control, and compliance you need to rapidly scale and respond to market and regulatory requirements, while controlling costs and improving your margins. With years of life sciences industry knowledge and experience, Armanino can help your company meet the complex operational and compliance challenges of your industry.
ArmaninoLLP is one of the largest Microsoft Dynamics partners in the nation as a value-added reseller (VAR) and implementation partner for Microsoft Dynamics AX, GP and CRM. Additionally, the firm offers the Microsoft Dynamics AX for High Tech Industries™ Certified for Microsoft Dynamics (CfMD) solution. This proprietary product helps technology companies, including software & internet, semiconductor, medical device, pharmaceutical and electronics manufacturers, to empower their sales forces, cut costs and time-to-market, improve forecast accuracy, and manage distributor relationships and supply chains. As a Microsoft Inner Circle partner, Armanino is among the top 1% of partners in the world.
You can count on Armanino to think strategically, to provide the sound insights that lead to positive action. We address not just your immediate issues, but your underlying business challenges, as well — assessing opportunities, weighing risks, and exploring the practical implications of both your short- and long-term decisions.
When you work with us, we give you options that are fully aligned with your business strategy. If you need to do more with less, we will implement the technology to automate your business processes. If it’s financial, we can show you proven benchmarks and best practices that can add value company-wide. If the issue is operational, we’ll consult with your people about workflow efficiencies. If it’s compliance, we’ll ensure you meet the requirements and proactively plan to take full advantage of the changes at hand. At every stage in your company’s lifecycle, we’ll help you find the right balance of people, processes, and technology.
Download White Paper

